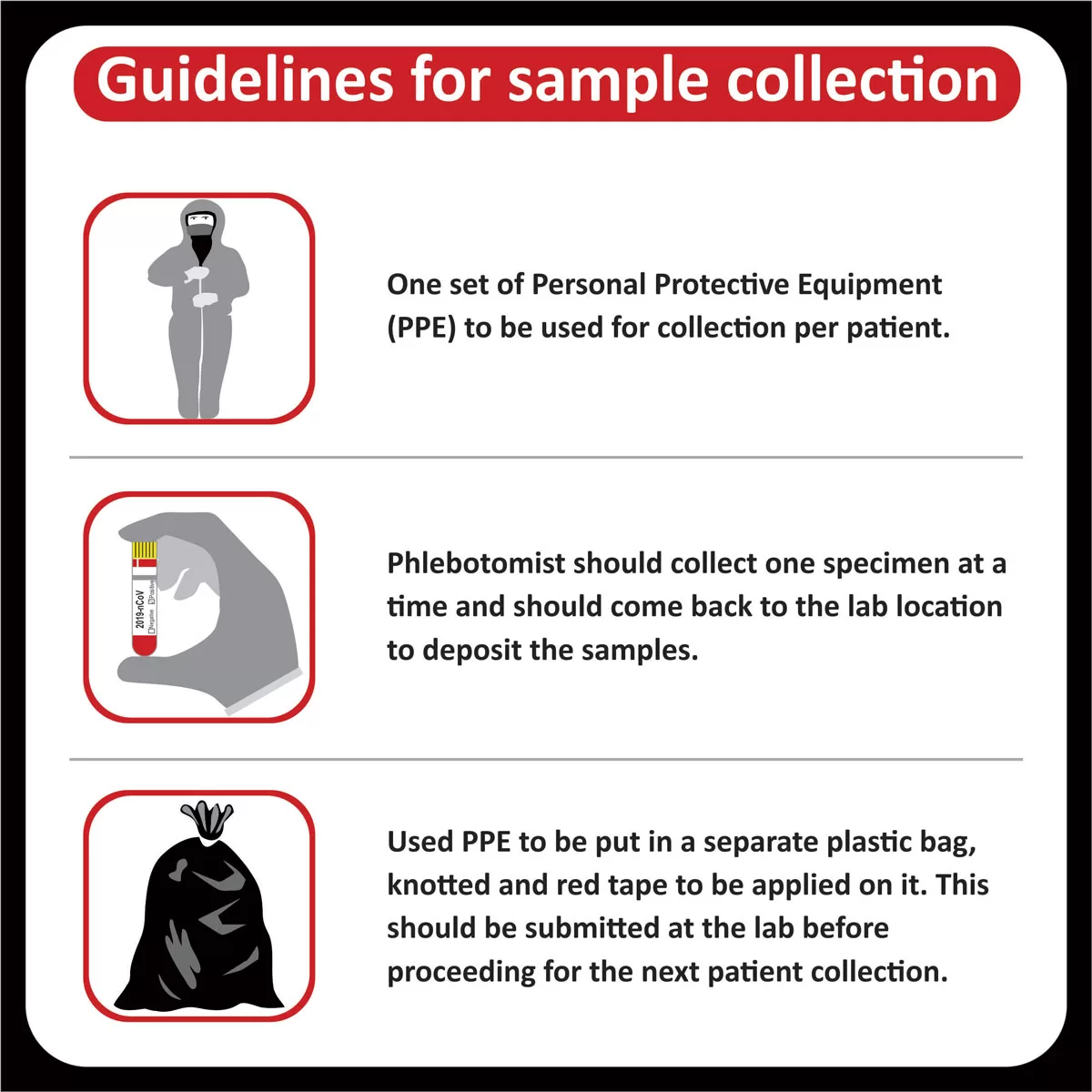
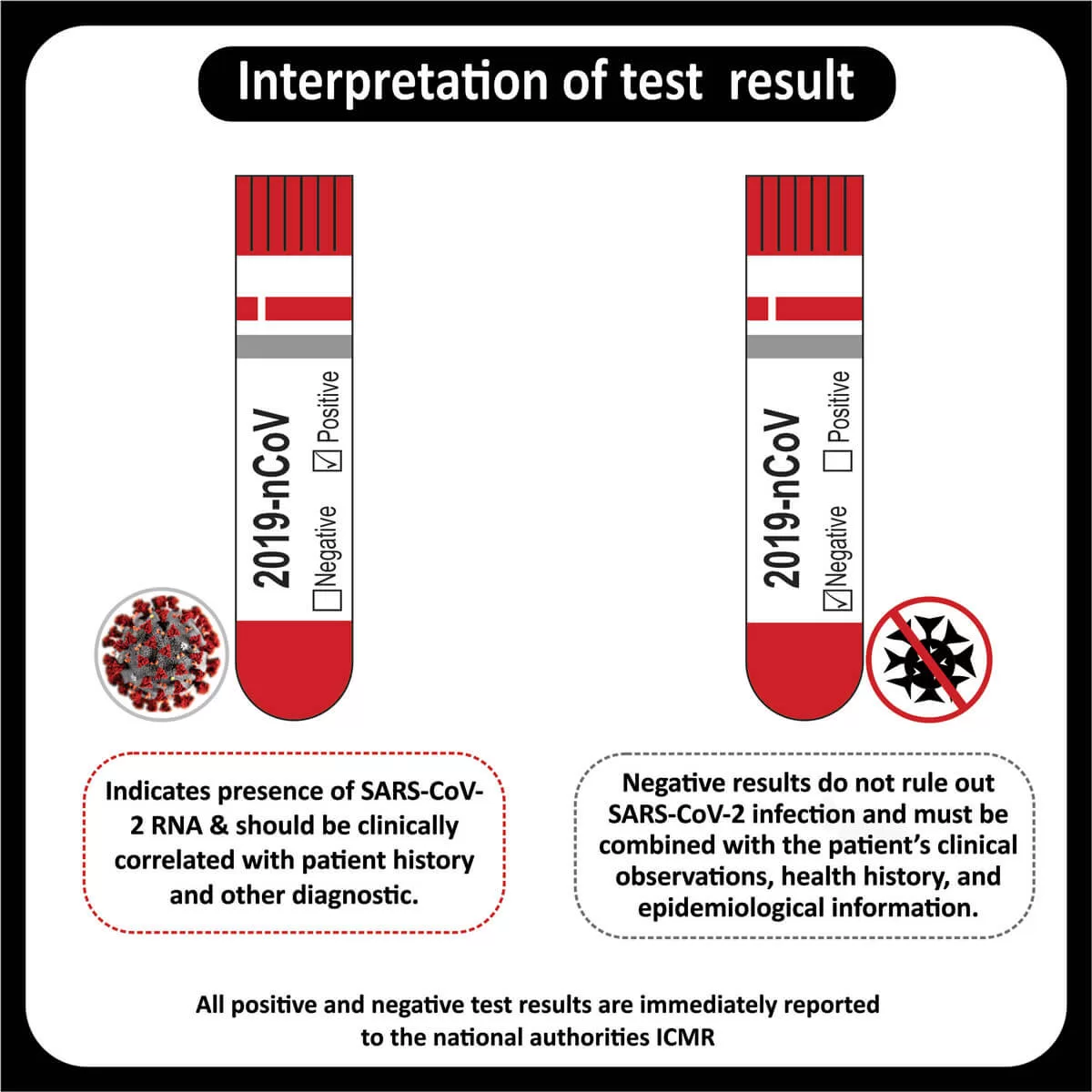
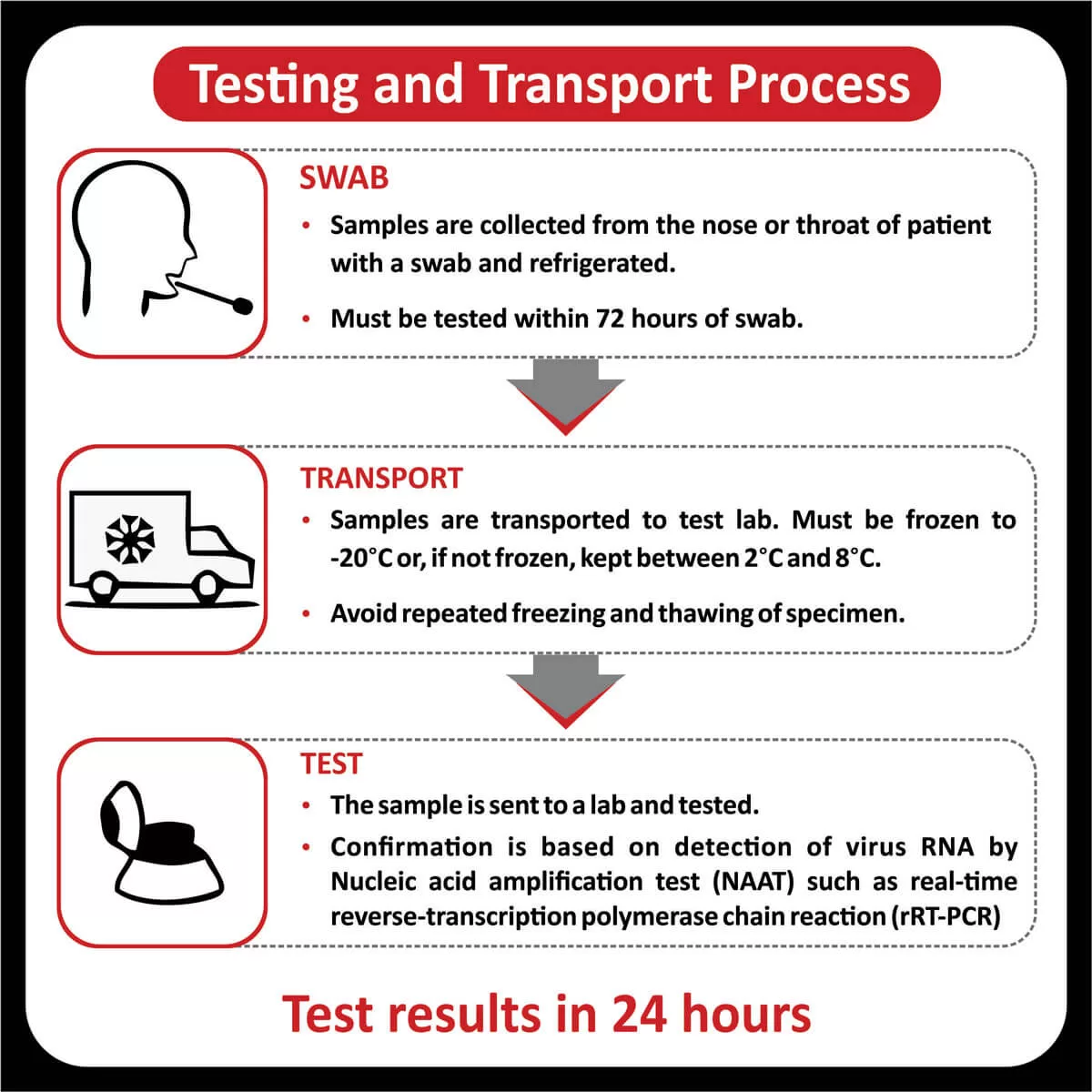
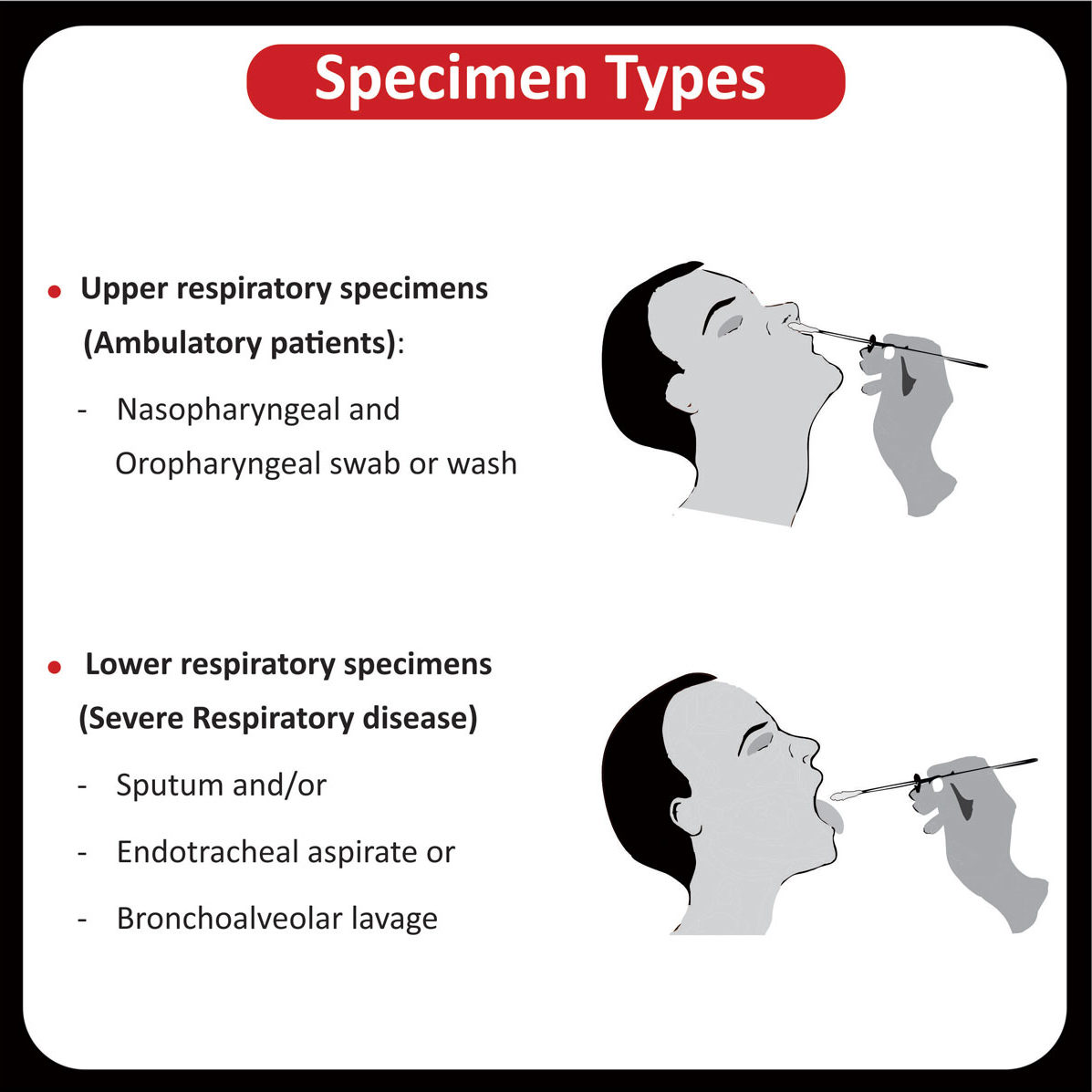
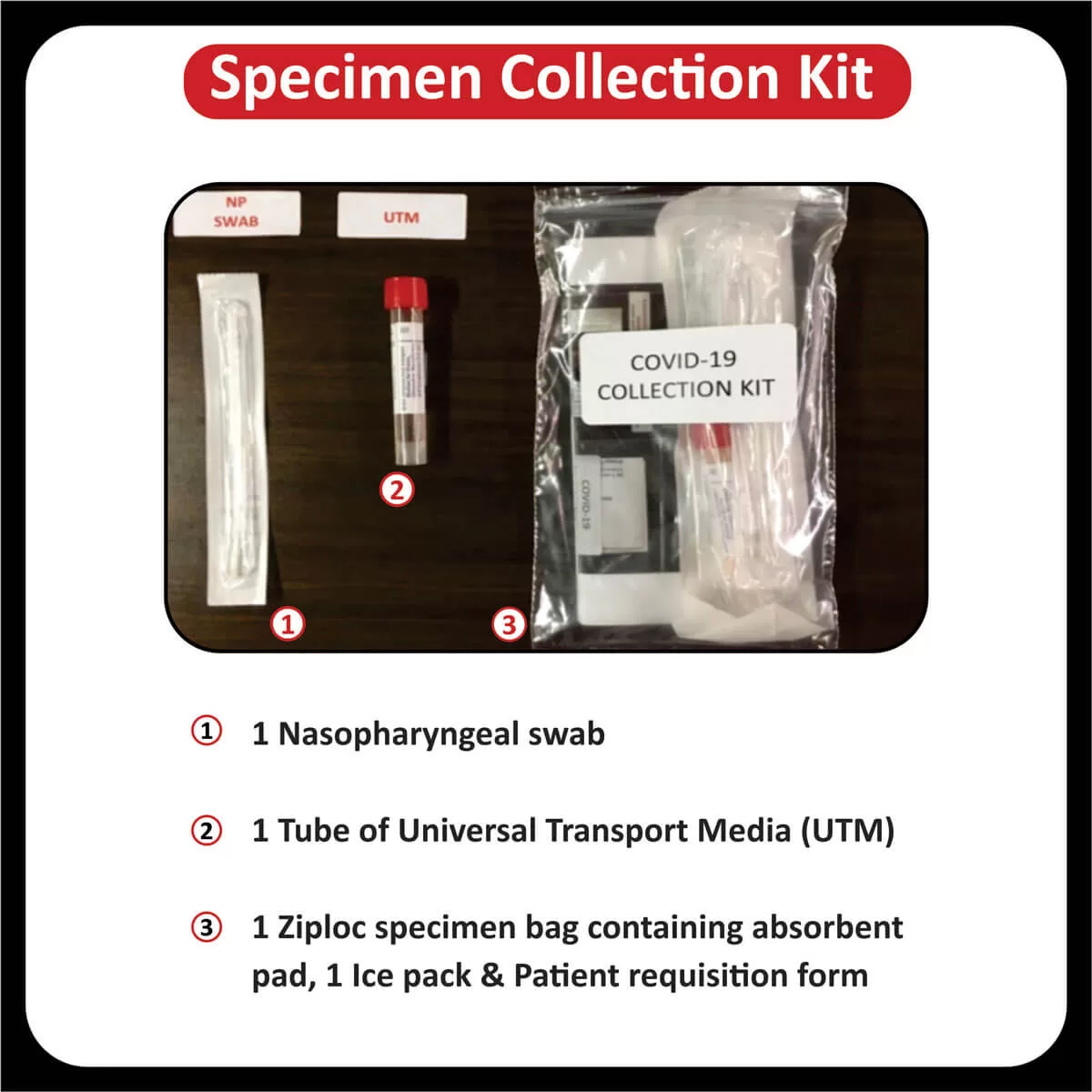
COREssence
The FDA granted accelerated approval to Atezolizumab (Tecentriq) plus nanoparticle albumin-bound (nab) Paclitaxel (Abraxane) for unresectable locally advanced...
In a recent clinical trial, researchers have found promising clinical activity of Pembrolizumab with Trastuzumab in patients with...
In a recent clinical trial, researchers found that patients with a higher tumor mutational burden, T cell inflamed...
The FDA approved Osimertinib (Tagrisso) for the first-line treatment of metastatic NSCLC with (EGFR) exon 19 deletions or...
The FDA granted approval to Gilteritinib for the treatment of adult patients who have relapsed or refractory acute...
A prospective cohort study of 323 patients with metastatic NSCLC showed marked increase in detection of therapeutically targetable...
The targeted therapy Venetoclax (Venclexta) can now be used for a broader group of patients with chronic lymphocytic...
The targeted therapy Venetoclax (Venclexta) can now be used for a broader group of patients with chronic lymphocytic...
The US Food and Drug Administration (FDA) has approved Pembrolizumab (Keytruda) for patients with recurrent or metastatic cervical...
The US Food and Drug Administration (FDA) has approved simultaneous administration of Tafinlar (Dabrafenib) and Mekinist (Trametinib) for...